 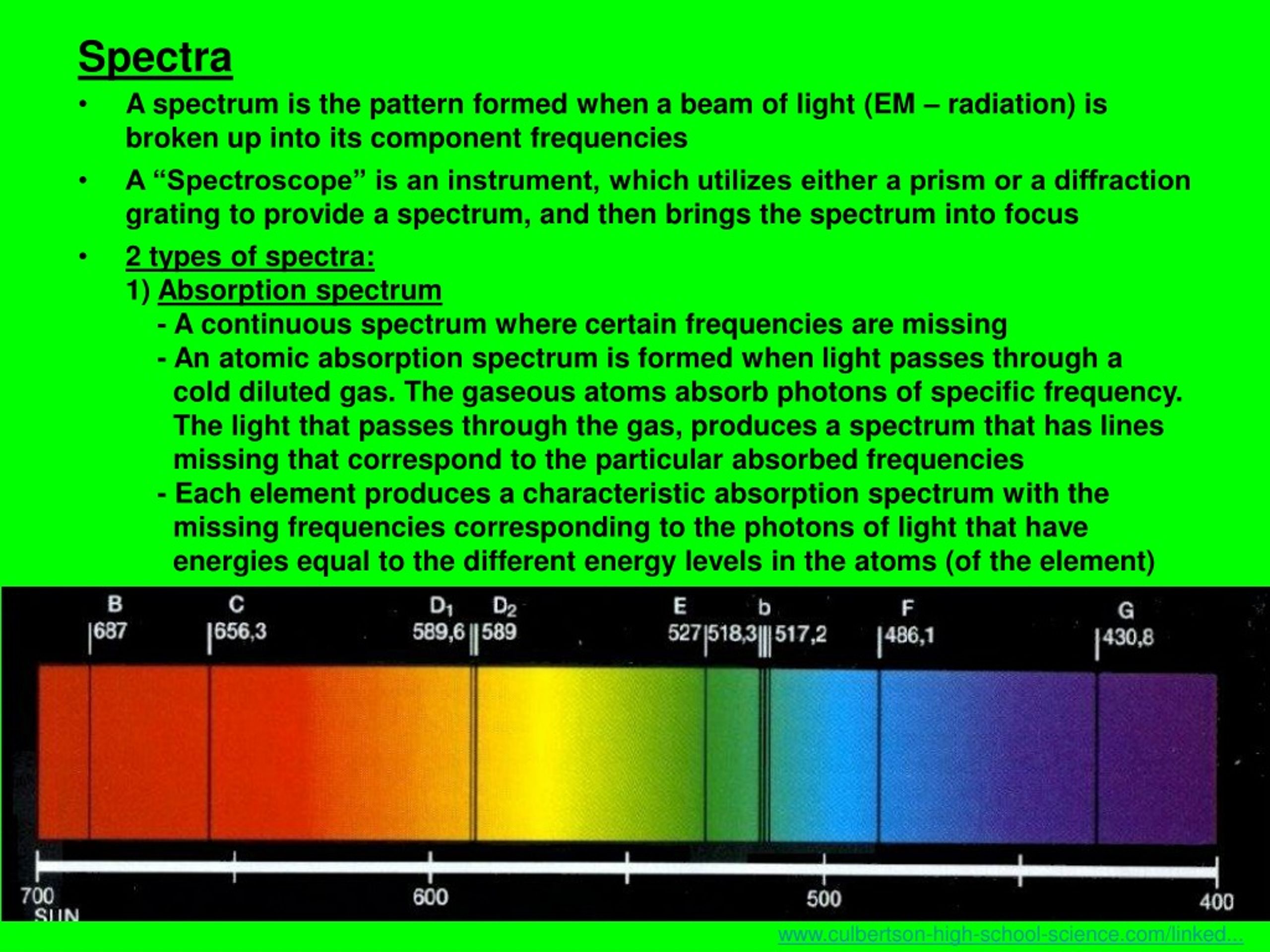
It ranges from the longest radio waves to the shortest X-rays and gamma rays. Rutherford’s earlier model of the atom had also assumed that electrons moved in circular orbits around the nucleus and that the atom was held together by the electrostatic attraction between the positively charged nucleus and the negatively charged electron. The meaning of ATOMIC SPECTRUM is a spectrum of radiation due to electron transitions within atoms and consisting mainly of series of spectrum lines. Spectrum: Definition The spectrum is a continuous range of electromagnetic radiation waves. Understand the processes of absorption and emission. Know that radiation of short wavelength is toxic. Understand that ionizing radiation can knock electrons off DNA. Bohr’s model required only one assumption: The electron moves around the nucleus in circular orbits that can have only certain allowed radii. Elizabeth Gordon Furman University Learning Objectives Differentiate between a continuous and discontinuous spectrum Classify all types of radiation as being ionizing or nonionizing. In 1913, a Danish physicist, Niels Bohr (1885–1962 Nobel Prize in Physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum. Define the ground state ground state and excited state of electrons Explain the difference between line spectra for absorption and emission of electrons. There are three kinds of atomic spectroscopy, they are. Where \(n_1\) and \(n_2\) are positive integers, \(n_2 > n_1\), and \( \Re \) the Rydberg constant, has a value of 1.09737 × 10 7 m −1. Atomic spectroscopy is a study of electromagnetic radiation that is absorbed or released from atoms. atomic spectrum synonyms, atomic spectrum pronunciation, atomic spectrum translation, English dictionary definition of atomic spectrum. \): When light from a hydrogen gas discharge tube is passed through a prism, the light is split into four visible lines.\] A spectral line is a weaker or stronger region in an otherwise uniform and continuous spectrum, resulting from emission or absorption of light in a narrow frequency range, compared with the nearby frequencies. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
